Environmental Science: The Pros and Cons of Ozone (Part 2)
Last time, we explored how stratospheric ozone shields Earth’s life from ultraviolet radiation. Today, we’ll examine the effects of ‘bad ozone’.
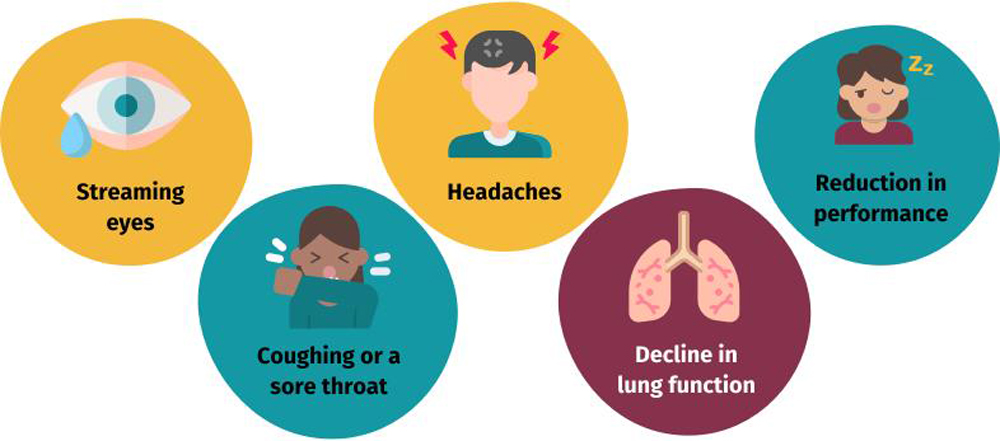
The term “harmful ozone” primarily refers to tropospheric ozone, specifically near-surface ozone located at altitudes of 10 to 100 meters above the ground. Elevated concentrations of near-surface ozone can exert detrimental effects on human health and the ecological environment. Studies indicate that children, women, the elderly, and individuals with pulmonary diseases (e.g., asthma) are particularly susceptible to the health hazards associated with ozone exposure. High ozone concentrations can irritate the eyes and respiratory tract, potentially impair pulmonary function, and simultaneously disrupt the healthy growth of plants.
When did ozone pollution appear in China?
The global ozone pollution issue was first identified by scientists in 1943 during the Los Angeles photochemical smog event. Photochemical smog refers to a harmful light blue haze formed when primary pollutants such as volatile organic compounds (VOCs) and nitrogen oxides (NOx) emitted from vehicles and factories undergo photochemical reactions under sunlight (ultraviolet light), generating secondary pollutants that subsequently mix with primary pollutants. The characteristic pollutant in this process is ozone, a potent oxidizing agent.
With the rapid development of China’s petrochemical industry, signs of photochemical smog pollution were first detected in 1974 in Xigu District, Lanzhou. Subsequent investigations conducted between 1976 and 1980 confirmed that the area was frequently shrouded in smog during clear summer days, causing residents to experience eye and respiratory irritation, while plants suffered significant damage. This marked the gradual emergence of ozone pollution concerns in the awareness of both Chinese scientists and the general public.

How is ground-level ozone formed?
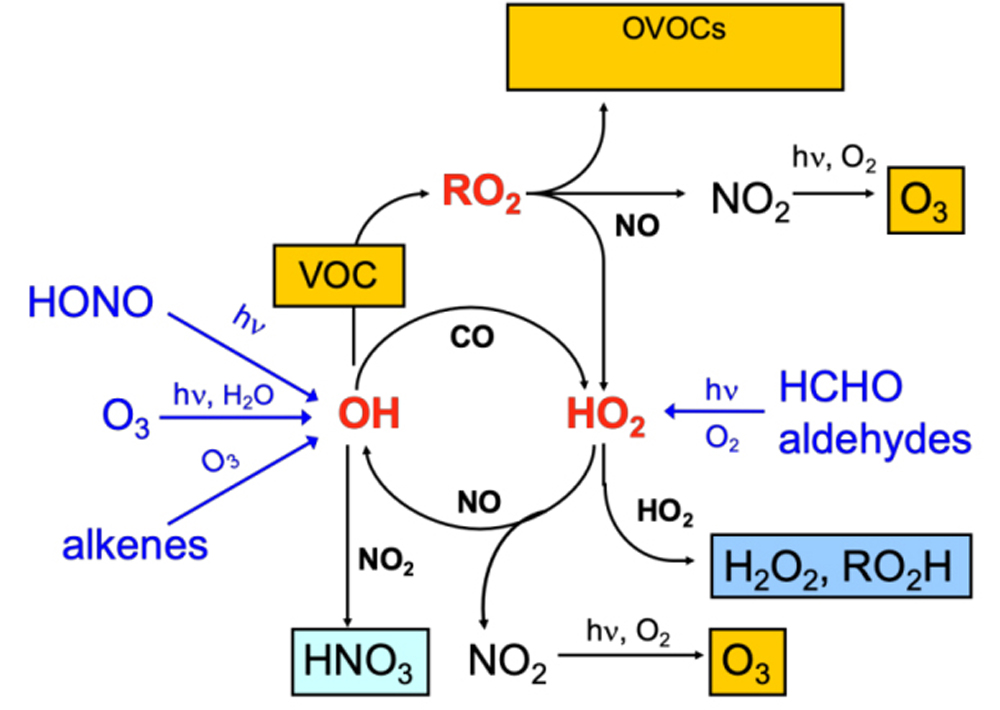
After nearly 40 years of scientific exploration and pollution prevention practices, Chinese researchers have reached a basic consensus on the formation mechanisms and control strategies of ozone pollution. Ozone in the near-surface atmosphere is primarily generated through a series of complex photochemical reactions, meaning that “bad ozone” is not directly emitted into the air but rather transformed from other pollutants.
The two main conditions for the formation of “bad ozone” are: ① the presence of precursor pollutants such as NOx and VOCs; ② intense sunlight (ultraviolet radiation).
1. Nitrogen Oxides (NOx) Emissions
Nitrogen oxides (NOx) are compounds composed of nitrogen and oxygen, including common types such as nitric oxide (NO), nitrogen dioxide (NO2), and nitrous oxide (N2O). While their sources are diverse, most emissions are anthropogenic, primarily stemming from fossil fuel combustion, industrial processes, and vehicle exhaust. Key industries and operations involved include thermal power generation, cement production, glass manufacturing, boiler combustion, industrial kilns, and transportation.
2. Volatile Organic Compounds (VOCs) Emissions VOCs, short for “Volatile Organic Compounds,” are categorized into 12 major types: benzene series, alkanes, alkenes, halogenated hydrocarbons, alcohols, ketones, phenols, aldehydes, ethers, esters, acids, and amines. Common VOCs include benzene, toluene, xylene, styrene, and trichloromethane. Compared to NOx, VOCs have a broader range of sources. Industrial enterprises in sectors such as petrochemicals, pharmaceuticals, chemicals, furniture manufacturing, automotive repair, and printing are typical sources of VOCs emissions. Additionally, VOCs are emitted from motor vehicles, gas stations, cooking fumes, and solvent emissions from dry cleaning services.
3. Light Conditions Summer is the peak season for ozone pollution, affecting not only China but also Europe and America. During summer, intense sunlight, high temperatures, low cloud cover, and strong ultraviolet radiation create ideal conditions for ozone formation. This explains why ozone concentrations peak in the late afternoon. Ozone levels typically rise from May, reach their maximum in July and August, and gradually decline as autumn sets in.
What is the current status of ozone pollution in China?
China’s Air Quality Index (AQI) is composed of six components: fine particulate matter (PM2.5), particulate matter (PM10), ozone (O3), sulfur dioxide (SO2), nitrogen dioxide (NO2), and carbon monoxide (CO). As a conventional air pollutant, ozone differs from SO2 and NOx—whose pungent odors are detectable—or PM2.5, which causes haze pollution, as it is neither “smellable” nor “visible.” By the end of the 12th Five-Year Plan period, China had established a national environmental air quality monitoring network to track real-time ozone data at monitoring stations. Additionally, in accordance with the Technical Specification for Environmental Air Quality Assessment (Trial) (HJ 663—2013), China employs the 90th percentile of the 8-hour moving average daily ozone concentration (MDA8-90) for annual ozone assessment.
In May this year, the Ministry of Ecology and Environment of China released the “2022 China Ecological Environment Status Bulletin”. According to the bulletin, in 2022, the 90th percentile concentration of the maximum 8-hour average daily O3 in 339 cities ranged between 90 and 194 micrograms per cubic meter, with an average of 145 micrograms per cubic meter, an increase of 5.8% compared to 2021. From a spatial distribution perspective, ozone concentrations in the southeastern coastal regions were often higher than those in inland areas.
Despite the significant challenges currently faced in ozone pollution control in China, the government has implemented a series of key measures in recent years, including the formulation and release of the Ozone Pollution Prevention and Control Action Plan, to further improve air quality and safeguard public health. With the collective efforts of all, we are confident that we will overcome the harmful effects of ozone, breathe cleaner air, and enjoy a happier life!
References :
[1] Institute of Physics and Chemistry, Chinese Academy of Sciences. The controversial ozone [EB/OL]. 2015-03-04. https://www.cas.cn/kx/kpwz/201503/t20150304_4317112.shtml
[2] China Atmospheric Ozone Pollution Prevention and Control Blue Book [S]. China Environmental Science Society Ozone Pollution Control Professional Committee. 2020-10.
[3] Chen CH, Huang JG, Ren ZH, et al. Meteorological conditions of photochemical smog pollution in Lanzhou Xigu Industrial Zone during summer [J]. Journal of Environmental Science, 1986(03):334-342. DOI: 10.13671/j.hjkxxb.1986.03.010.
[4] Ministry of Ecology and Environment of the People’s Republic of China, 2022 China Ecological Environment Status Bulletin [EB/OL]. https://www.mee.gov.cn/hjzl/sthjzk/zghjzkgb/202305/P020230529570623593284.pdf.
Source: Da Cheng Xiao E
